Nickel Titanium Alloy(nitinol smart alloy)
Nickel–titanium alloy, or Nitinol alloy, is nitinol smart alloy. It is nitinol a shape memory alloy.why is nitinol called a smart alloy? 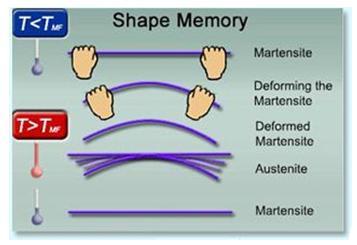

Industrial processing of titanium–nickel (Ti–Ni) shape memory alloys (SMAs) to achieve key properties
T. Nakahata, in Shape Memory and Superelastic Alloys,2011
1,Introduction
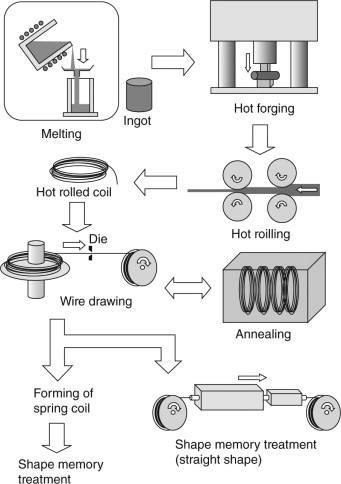
Titanium–nickel (Ti–Ni) alloys are widely used as the more practical shape memory or superelastic alloys due to their excellence of shape recover-ability, durability and corrosion resistance. Ti–Ni alloys also exhibit excellent workability compared with other shape memory alloys (SMAs) although cold workability is poor when compared with conventional metallic materials such as steel. As shown in Fig. 4.1, the fabrication process of the Ti–Ni alloys consists of the several steps of melting, hot and cold working, forming and shape memory treatment. The fabrication process is similar to those used for other metallic materials, but there are some differences. Firstly, to draw wires, soft annealing must be repeated until the required dimensions are attained since the cold workability of the material is poor. Secondly, shape memory treatment (heat treatment) is required in the final stage of the process. It is known that each processing step affects the characteristics of the Ti–Ni alloys such as transformation temperatures, durability and elastic modulus. Industrial processing techniques for Ti–Ni SMAs and the methods for controlling their properties are described in this chapter.
2,Biomaterials and Applications
Maria Cristina Tanzi, Gabriele Candiani, in Foundations of Biomaterials Engineering,2019
2.1 Ni-Ti Alloy
Ni-Ti alloy (also known as Nitinol) is an alloy with a near-equiatomic composition (i.e., 49%–51%) of nickel and titanium. Ni-Ti belongs to the class of shape memory alloys that can be deformed at a low temperature and are able to recover their original, permanent shape when exposed to a high temperature. The shape memory effect of Nitinol is related to its martensitic transformation. In fact, at high temperature, the atoms are arranged in an orderly manner, in a BCC crystal structure (i.e., austenitic phase). In this phase, the alloy is resistant to twisting and bending. At low temperature, Ni-Ti alloy exhibits a monoclinic distorted crystal structure, that is, a martensitic phase. At this stage, the alloy can be easily deformed. The temperature at which the austenitic phase transforms into a martensitic one is called the transformation temperature. The atomic percentage composition (Table 4.7) is important in determining the shape memory effect at body temperature; in fact, the presence of a higher content of nickel (even by 0.1%) can induce a decrease in the transformation temperature and an increase in yield strength of the alloy. Moreover, contaminants, such as carbon and oxygen, can vary the transformation temperature, affecting the mechanical properties. Hence, attention should be taken to minimize the concentration of the contaminants.

3,Shape memory, superelastic and low Young's modulus alloys
M. Niinomi, in Biomaterials for Spinal Surgery,2012
3.1Abstract:
The conventional shape memory alloy, TiNi, is widely used in biomedical implants. TiNi is receiving particular attention owing to its usefulness in stents and guide wires for catheters. However, the Ni in TiNi poses a high risk of allergic reaction, and so Ni-free shape memory and superelastic titanium alloys are being developed. Titanium alloys with a low Young’s modulus are also required to inhibit stress shielding between implant and bone, because stress shielding leads to bone atrophy and poor bone remodeling. Titanium alloys with adjustable Young’s moduli are being developed in order to satisfy the requirements of both surgeons and patients. In this chapter, the fundamental characteristics of shape memory and superelastic alloys for biomedical applications are discussed. In addition, the effects of alloying elements on the shape memory, superelastic behavior, biocompatibility and corrosion resistance, and endurance of TiNi are described. Further, the fabrication of TiNi sheets is covered. Finally, the developments of a new alloy, low Young’s modulus alloys, and the metals required for spinal surgery are discussed.

4,Metals and alloys for biomedical applications
Mehar Al. Minnath, in Fundamental Biomaterials: Metals,2018
4.1Nickel–titanium alloys
Nickel–titanium alloy, or Nitinol, is a smart material with shape memory and superelastic properties. It has an elastic modulus closer to that of bone than other metal and metal alloy implant materials. Nitinol finds application in orthodontics, in the treatment of bone fractures, and as bone suture anchors for attaching soft tissues such as tendons and ligaments to bone. Because of its shape memory property, NiTi also are used as stents for reinforcement of blood vessels. As any other Ti alloy, NiTi displays excellent biocompatibility due to the formation of a thin titanium oxide surface. But unlike ather alloys containing Ni, Nitinol is highly stable in the body with minimal Ni release. Introduction of Cu to Nitinol (TiNiCu) has attracted interest for biomedical and other applications due to better fatigue and modified shape memory properties
5,Blood flow dynamics and surface interactions
W VAN OEVEREN, in Surfaces and Interfaces for Biomaterials,2005
5.1Nitinol
Nickel-titanium alloy (Nitinol) has attracted special attention due to its shape memory function. It must be noted that Nitinol has an outer surface of titanium (oxide), whereas nickel is not exposed to blood. Therefore, blood compatibility characteristics are expected to be rather similar to those of titanium oxide. Based on the hypothesis that a semi-conductor prohibits fibrin and platelet deposition, Nitinol is expected to be thromboresistant, unless its semi-conductive nature is lost in the alloy. In a clinical study with Nitinol intravascular clot filters, the effects on the clotting system and on platelet adhesion were shown to be similar to those induced by stainless steel.112 An experimental study with stented rabbits showed significantly more thrombus formation on stainless steel than on Nitinol.113 However, grafting of polyethylene oxide (PEO) on Nitinol reduced the fibrinogen adsorption by as much as 99%, and significantly reduced platelet adhesion, which once more shows the superior thromboresistant effects of polymers as compared to those of metals like Nitinol.114
Further evidence that Nitinol, too, can only be safely implanted during antithrombotic treatment was provided in experiments that included the use of platelet inhibitors Aspirin and Copidrogel in a porcine stent model. Combined treatment with these inhibitors reduced stent thrombosis by 95–98%.115 An effective coating such as PEO could thus limit the use of systemic treatment by medication.

